When you pick up a prescription, you might see two options: the familiar brand-name pill or a much cheaper generic version. Many people wonder-are generic drugs as safe as brands? The answer isn’t as simple as yes or no, but the clinical evidence paints a clear picture: for the vast majority of medications, generics work just as well and are just as safe.
What makes a drug "generic"?
A generic drug isn’t a copycat or a knockoff. It’s required by law to contain the exact same active ingredient, in the same strength, same dosage form (pill, injection, capsule), and same route of administration (oral, topical, etc.) as the brand-name version. The U.S. Food and Drug Administration (FDA) doesn’t just approve generics based on trust-they demand proof. That proof comes from bioequivalence studies, which measure how quickly and completely the body absorbs the drug. The standard? The generic must deliver the same amount of medicine into your bloodstream within 80% to 125% of the brand-name drug’s levels. That’s not a guess. It’s science.
So why are generics cheaper? Because they don’t need to repeat expensive clinical trials. The original brand company already proved the drug works and is safe. Generic manufacturers only need to show their version behaves the same way in the body. This saves billions. In 2023, generics made up 90% of all prescriptions filled in the U.S., but only 18% of total drug spending. That’s how much money patients and insurers save.
Does the science back it up?
A 2020 study published in Nature Scientific Reports looked at over 1.3 million patients in Austria who took either brand or generic versions of 17 different drugs, including heart medications, antidepressants, and blood thinners. Researchers compared death rates and serious heart and stroke events. The results? Generic drugs were linked to fewer deaths in 10 of the 17 drugs. For example, patients on generic versions of beta-blockers and statins had 40.6 deaths per 1,000 patient-years, while those on brand-name versions had 45.8. That’s not a fluke. The study ruled out random chance and even considered whether doctors might have prescribed generics to sicker patients. The results held.
Another large study from the U.S. analyzed over 136,000 seniors in Canada after generic versions of three common blood pressure drugs-losartan, valsartan, and candesartan-were introduced. In the month after generics hit the market, there was an 8% to 14% increase in hospital visits and emergency care for high blood pressure problems. At first glance, this seems to suggest generics are less safe. But here’s the catch: this spike likely happened because pharmacists automatically switched patients to generics without checking if their doctor had approved it. Some patients had been stable on their brand-name drug for years. Switching them without warning, without monitoring, and without adjusting doses created instability. The problem wasn’t the generic-it was the switch.
When might generics cause problems?
There are a few drugs where tiny differences in how the body absorbs the medicine can matter. These are called narrow therapeutic index drugs. The difference between a dose that works and one that’s toxic is very small. For these, even a 5% variation in absorption can be risky.
Examples include:
- Levothyroxine (for thyroid disease)
- Warfarin (a blood thinner)
- Phenytoin and other anti-seizure medications
- Cyclosporine (for organ transplant patients)
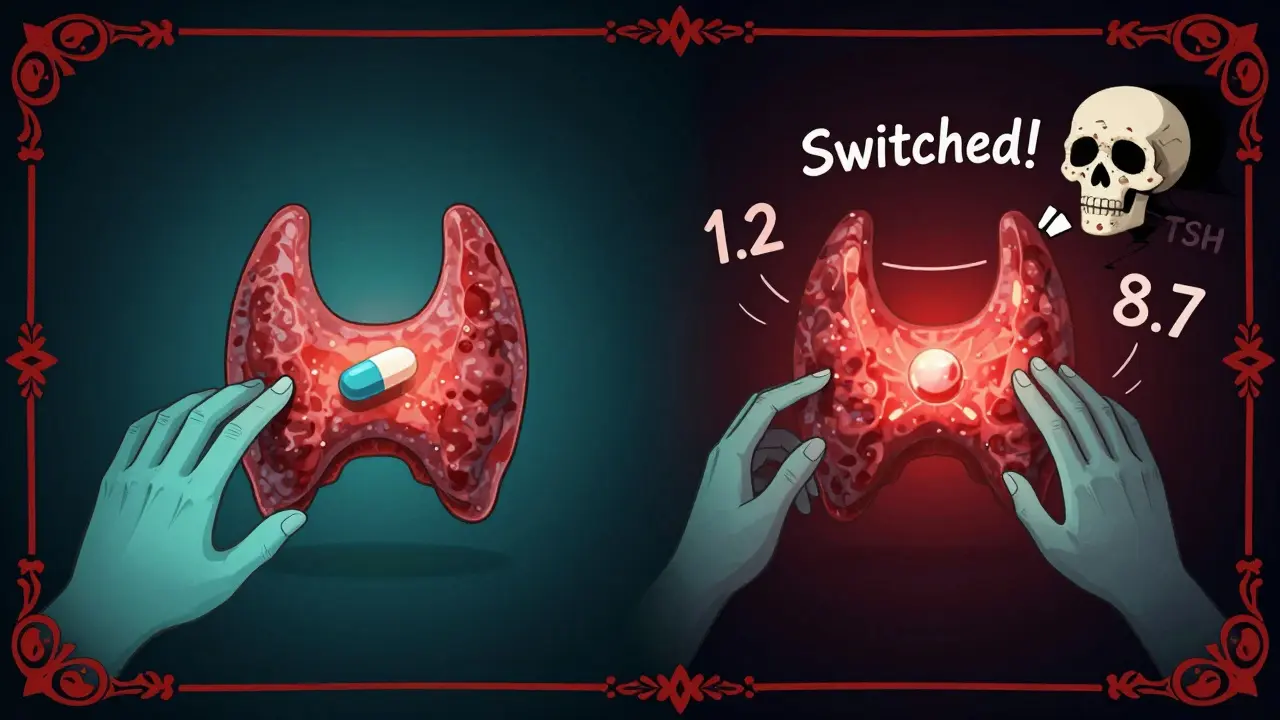
Some patients report feeling worse after switching from brand to generic for these drugs. One Reddit user shared that after switching from Synthroid (brand) to generic levothyroxine, their TSH level jumped from 1.2 to 8.7-signaling their thyroid wasn’t being controlled anymore. That’s not rare. A 2021 survey found 19% of thyroid patients reported side effects or loss of symptom control after switching generics. The American Thyroid Association recommends sticking with one version-either brand or generic-and avoiding switches unless absolutely necessary.
Why does this happen? It’s not because generics are low quality. It’s because of inactive ingredients. Generics can use different fillers, dyes, or coatings. For most people, this doesn’t matter. But for someone with a rare allergy or a sensitive digestive system, even a small change in excipients can affect how the drug dissolves or is absorbed.

What do real-world users say?
On Drugs.com, 89% of users who took generic versions of common drugs like atorvastatin (for cholesterol) or metformin (for diabetes) reported no difference from the brand. But 11% said they noticed something off-usually with psychiatric meds or seizure drugs. Pharmacists in the U.S. report the same pattern: most patients do fine, but a small group insists they feel worse on generics.
The FDA’s own adverse event database, FAERS, shows no meaningful difference in reported side effects between brand and generic versions of 15 common drugs. For every 1 million prescriptions, generics had 12.7 reports of side effects; brands had 13.2. That’s not a gap-it’s noise.
So why do some people swear generics don’t work for them? Sometimes, it’s the placebo effect in reverse. If you believe a brand-name drug is better, your body might respond accordingly. Other times, the switch itself disrupts a routine. If you’ve been taking the same pill for years, changing the shape, color, or size-even if it’s chemically identical-can trigger anxiety. And anxiety can worsen symptoms.
What should you do?
If you’re on a drug with a narrow therapeutic index-like levothyroxine or warfarin-talk to your doctor. Ask if you should stick with one version. Don’t let your pharmacy switch you without warning. You have the right to request the brand if you’ve had issues.
For most other medications-antibiotics, blood pressure pills, antidepressants, statins-generics are safe, effective, and a smart financial choice. The data is overwhelming: they work just as well.
If you switch and feel worse, don’t assume it’s the drug. Track your symptoms. Keep a log: when did you switch? What changed? Did your dosage change? Did your sleep, appetite, or energy shift? Bring this to your doctor. Sometimes, a small dose tweak fixes the problem. Other times, you might need to go back to the brand.
Who decides what gets switched?
In most states, pharmacists can automatically substitute a generic unless the doctor writes "Dispense as Written" on the prescription. But many doctors don’t check the FDA’s Orange Book-where drugs are rated for therapeutic equivalence-before prescribing. One study found only 43% of physicians regularly check these ratings. That means switches happen without informed consent.
Patients should know: you can ask for the brand. You can ask to stay on the same version. You can ask your pharmacist to notify you before switching. You have a voice in this.
Is there a catch?
Yes. Not all generic manufacturers are created equal. The FDA inspects over 3,000 drug manufacturing facilities worldwide. In 2022, they issued 17 import alerts for facilities producing generics due to quality issues-contaminated ingredients, poor record-keeping, or unapproved processes. Most of these were in India and China. But here’s the key: if a batch fails inspection, it’s pulled. The FDA doesn’t let unsafe drugs hit shelves. And if a generic fails bioequivalence testing, it’s rejected before it ever gets to you.
So while manufacturing quality matters, the system is designed to catch problems before they reach patients. The risk isn’t zero-but it’s extremely low.
Bottom line
For 90% of prescriptions, generic drugs are just as safe and effective as brand-name drugs. The science backs it. The numbers back it. Real-world data backs it. The exceptions exist, but they’re rare and specific. If you’re on a drug where small changes matter-thyroid, seizure, or blood thinner-talk to your doctor. Otherwise, save money without sacrificing safety. Generics aren’t a compromise. They’re a smart, science-backed choice.
Are generic drugs as safe as brand-name drugs?
Yes, for the vast majority of medications. The FDA requires generics to contain the same active ingredient, strength, dosage form, and route of administration as the brand. They must also prove bioequivalence-meaning they deliver the same amount of medicine into your bloodstream within a strict range (80-125%). Large studies, including one published in Nature Scientific Reports, show generics are just as safe and sometimes even linked to fewer adverse events.
Can generic drugs cause side effects that brand drugs don't?
Generally, no. The active ingredient is identical, so the core side effects are the same. However, generics can use different inactive ingredients (fillers, dyes, coatings), which may cause reactions in rare cases-like allergies or digestive upset. Some patients report feeling worse after switching, especially with thyroid, seizure, or blood thinner medications. This isn’t because the generic is unsafe, but because even small changes in absorption can affect people with narrow therapeutic windows.
Why do some people say generics don't work for them?
Sometimes it’s psychological-switching from a familiar pill to a different-looking one can trigger anxiety. Other times, it’s a real physiological response. For drugs like levothyroxine or warfarin, even slight differences in absorption can affect how well the drug works. A patient might switch from brand to generic and notice their symptoms return. In these cases, going back to the original version often helps. It’s not that generics are inferior-it’s that consistency matters more for these specific drugs.
Should I avoid generic drugs if I have a chronic condition?
Not necessarily. For most chronic conditions-like high blood pressure, diabetes, or high cholesterol-generics are perfectly safe and effective. But if you’re on a drug with a narrow therapeutic index (like levothyroxine, warfarin, or phenytoin), ask your doctor whether you should stick with one version. Avoid switching back and forth. If you’ve been stable on a brand, you may want to request that your prescription be marked "Dispense as Written."
How can I tell if a drug is generic or brand?
The packaging and pill imprint will look different. Generics are labeled with the drug’s chemical name (like "atorvastatin") instead of a brand name (like "Lipitor"). The pill may be a different color, shape, or size. Your pharmacy label will list the manufacturer. If you’re unsure, ask your pharmacist. You can also check the FDA’s Orange Book online to see if a generic is rated as therapeutically equivalent to the brand.
Do pharmacists have to tell me if they're switching my drug to generic?
In most U.S. states, pharmacists can substitute a generic without telling you-unless the doctor wrote "Dispense as Written" on the prescription. But you have the right to ask. Many pharmacies will notify you if they’re making a switch. If you’re concerned, always ask: "Is this the same as my usual medication?" and "Can I request the brand if needed?"